Conventional microarrays are made by adsorbing antibodies onto hydrophobic or chemically reactive solid surfaces, which can lead to denaturation, random orientation, loss of binding activity and reduced assay sensitivity. To address these challenges, Wold et al. (ref. 1) developed a microarray platform that leverages site-specific antibody–oligonucleotide conjugates.
Antibodies expressed in E. coli are engineered to incorporate an unnatural amino acid, p-acetylphenylalanine (pAcF), using a nonsense amber codon and an orthogonal amber suppressor aminoacyl tRNA synthetase/tRNA pair specific for pAcF. The ketone group of the pAcF residue undergoes a bioorthogonal reaction with single-stranded aminooxy-modified oligonucleotides to form a stable oxime linkage.
Reactions were carried out in the presence of 100 mM methoxy-aniline catalyst at pH 4.5 and 37 degrees for 16 hours, followed by Mono Q anion-exchange chromatography. The production of a homogeneous, site-specifically labelled Fab–oligonucleotide conjugate was confirmed by SDS-PAGE.
The conjugated antibodies can then hybridise to complementary DNA sequences pre-printed on glass slides, anchoring the antibodies in a uniform orientation while preserving their antigen-binding functionality. The selectivity of antibody capture across different arrays was demonstrated using an anti-kappa phycoerythrin conjugate.
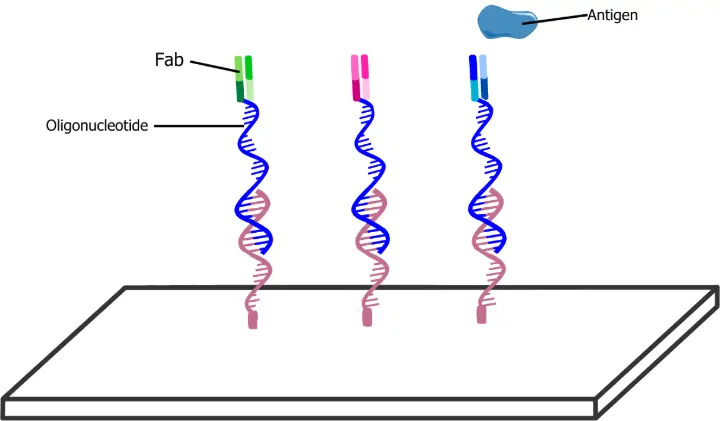
Wold et al. focused on detecting Her2-positive breast cancer cells (SK-BR-3) using Fab fragments of the therapeutic antibody trastuzumab, which was tagged with an oligonucleotide at a surface-exposed light chain site, S202. Modification here is known not to affect antigen-binding affinity or specificity.
The Fab-oligonucleotide conjugates were homogeneous, properly orientated after hybridisation, and retained their binding activity. HER2-positive cells selectively bound to the antibody-DNA arrays, whereas HER2-negative cells exhibited negligible binding. By cutting the double-stranded anchor with restriction enzymes, it was shown that capture depended on DNA hybridisation, highlighting the platform’s specificity.
The application of antibody-oligonucleotide conjugates along with carefully designed orthogonal capture sequences supports multiplexed detection of protein biomarkers or cells.